Nanostics Receives Funding from the University of Alberta Innovation Fund to Propel Adoption of the ClarityDX Prostate Test
perrin2024-04-23T12:23:50+00:00- University of Alberta Innovation Fund invests in Nanostics to support adoption of its ClarityDX Prostate test
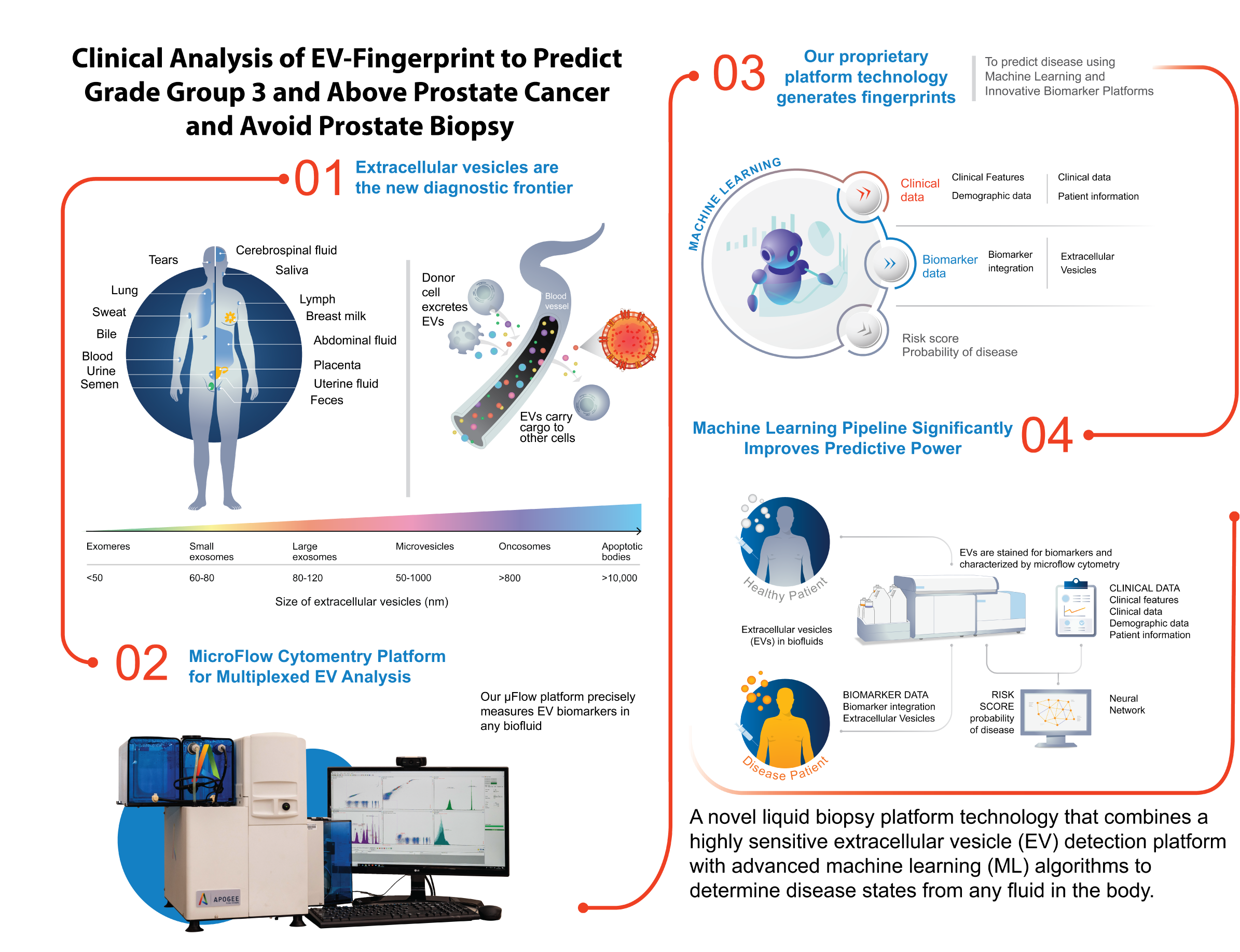
- ClarityDX Prostate is a blood test that combines biological and clinical information using machine learning to generate a risk score for aggressive prostate cancer
EDMONTON, ALBERTA – April 23 – Nanostics Inc., a precision health company developing diagnostic tests with its ClarityDX® platform technology, announced today that it received an investment from the University of Alberta Innovation Fund (UAIF) to help propel adoption of Nanostics’ ClarityDX Prostate® test in North America.
Funds raised through the UAIF will go towards expanding access to ClarityDX Prostate across North America and supporting the expansion of its product pipeline, including the development of ClarityDX Bladder, a minimally invasive bladder cancer diagnostic test.
ClarityDX Prostate is currently available in Canada as a laboratory-developed test. For more information or to order the test please visit nanosticsdx.com.
Click below to read the full press release.